For Research and Laboratory Use Only
 17% OFF
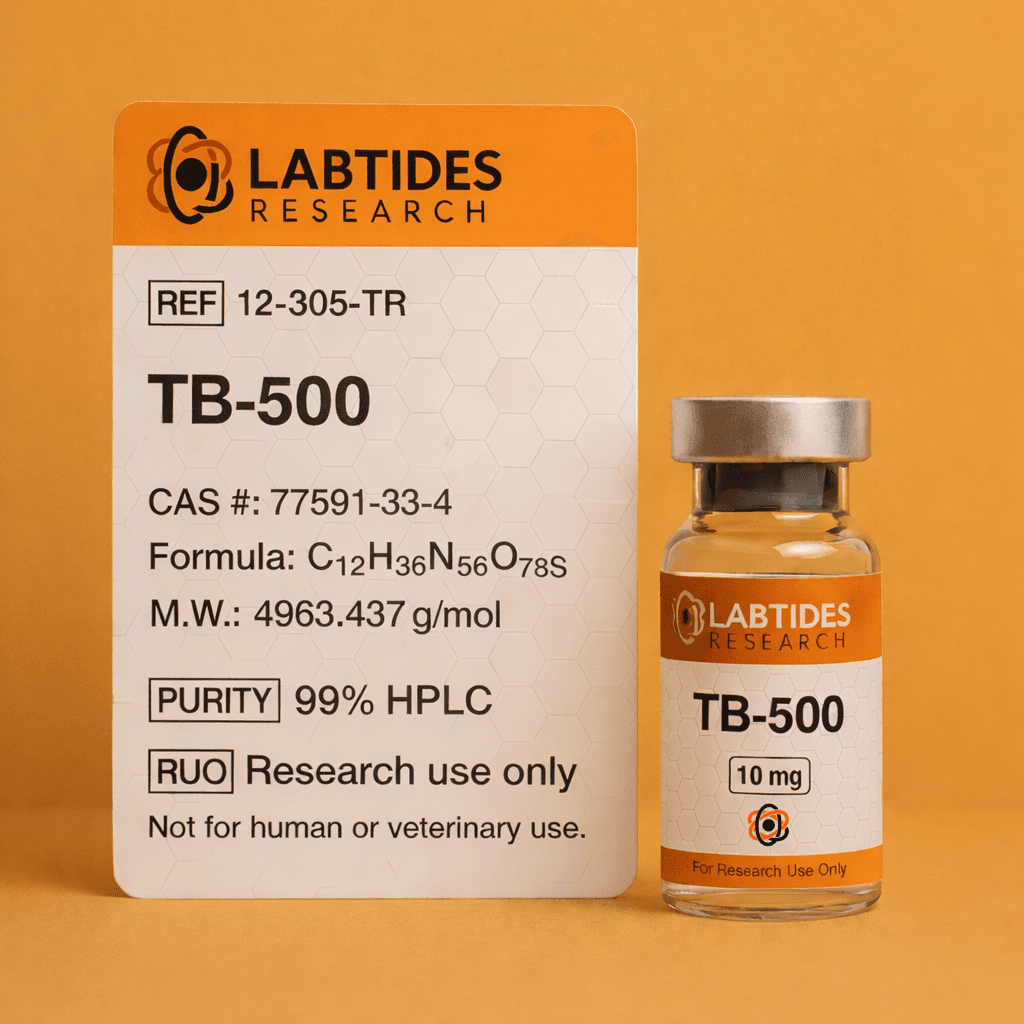
17% OFFThymosin Beta-4 (TB-500) is a naturally occurring peptide present in almost all human and animal cells. Research focuses on its role in cell migration and tissue repair mechanisms. 99% HPLC purity with full Certificate of Analysis. CAS #: 77591-33-4.
$59.99
$49.99
HPLC tested >99%
Mon, Jun 8 – Wed, Jun 10
For research and laboratory use only. Not for human consumption, therapeutic, or diagnostic use. Must be 21+ to purchase.
FASTER HEALING
Re-epithelialization vs controls at 7 days
SERIOUS ADVERSE EVENTS
Phase 1 trial with 42-1260mg IV doses
CITED STUDIES
Goldstein et al. review cited 170+ times
AMINO ACIDS
Naturally occurring peptide sequence
HALF-LIFE
Dose-dependent (42-1260mg IV)
MECHANISM OF ACTION
Thymosin Beta-4 (TB-500) is a naturally occurring 43-amino acid peptide that plays a vital role in tissue repair and regeneration. It sequesters G-actin monomers, promotes cell migration, inhibits inflammation, and stimulates angiogenesis.
Monomeric Globular Actin Binding
ILK/PINCH/Parvin Complex Activation
From clinical studies and research protocols
40 healthy volunteers, randomized, double-blind, placebo-controlled
Low dose cohort
t½ = 0.95 hours
Medium-low dose
t½ = 1.2 hours
Medium-high dose
Dose-proportional response
PHARMACOKINETICS
Half-life increases with increasing dose (dose-dependent kinetics)
Dose-proportional pharmacokinetic response observed
SAFETY PROFILE
FAQ
TB-500 is a synthetic version of a 43-amino acid peptide called Thymosin Beta-4. First identified in the thymus gland in the 1960s, it is present in virtually all cells and tissues, with high concentrations in blood platelets and wound fluid.
·Research compound — not approved for human use
·Banned by WADA (S0 category — non-approved substances)
·Phase 2 trials completed for dry eye disease
·Limited long-term safety data available
NF-κB and Toll-like Receptor Modulation
TB-500 binds to G-actin monomers, preventing premature polymerization and allowing coordinated cytoskeletal remodeling. This promotes efficient cell migration to injury sites.
Cell Migration
2-3x increase
Re-epithelialization (Day 7)
+61%
Wound Contraction
+11% more
Collagen Deposition
Increased
CLINICAL / RESEARCH RESULTS
A randomized, placebo-controlled Phase 1 trial demonstrated that IV Tβ4 at doses of 42-1260mg for 14 days was well tolerated with no dose-limiting toxicities in healthy volunteers.
3.75 mg/kg
Optimal dose
For neurological recovery (stroke model)
14 days
Treatment duration
Phase 1 multiple dose regimen
100%
Completion rate
No dropouts due to adverse events
RESEARCH APPLICATIONS
Accelerated wound closure and re-epithelialization in animal models. 61% faster re-epithelialization at 7 days.
Source: J Invest Dermatol
Cardioprotective effects studied in myocardial infarction models. Activates ILK, promotes cardiomyocyte survival.
Source: Nature (2004)
Improved functional outcome in stroke and TBI models. Promotes oligodendrogenesis and axonal remodeling.
Source: J Neurol Sci
Dry eye and corneal wound healing applications. Phase 2 trials completed for dry eye disease.
Source: Clin Ophthalmol
High dose cohort
t½ = 1.9 hours
·All doses well tolerated with no dose-limiting toxicities
·Half-life increased with increasing dose (0.95-1.9 hrs)
·Pharmacokinetic profile showed dose-proportional response
Optimal neurological dose calculated at 3.75 mg/kg
Low dose
Significant improvement (p<0.05)
Calculated optimal dose
Maximum predicted efficacy
Ceiling dose
No significant improvement (ceiling effect)
·Optimal dose calculated via quartic regression
·Ceiling effect observed at 18 mg/kg
·Treatment initiated 24 hours after injury
Naturally present in high concentrations in blood platelets
Because Tβ4 promotes cell migration and angiogenesis, there are theoretical concerns about potential effects on tumor growth and metastasis.
·Phase 1 data in 40 healthy volunteers showed excellent tolerability
·Animal toxicology: NOEL ≥18 mg/kg
·Salivation observed in dogs at higher doses (25-60 mg/kg)
COMPOUND INFORMATION
Lyophilized (powder)
Reconstituted